What is the PRRS-resistant pig?
PIC’s PRRS-Resistant Pig was developed to protect pigs from one of the most devastating swine diseases around the world, the Porcine Reproductive and Respiratory Syndrome (PRRS) virus. A recent study from Iowa State University reported that PRRS costs $1.2 billion per year, in the U.S. alone. That number has increased 80% from previous estimates of $664 million between 2006 – 2010 and $560 million between 2000 and 2005.
If we could eliminate the challenges from the most serious global pig disease, we could have pork with improved animal welfare, reduced need for antibiotics and a reduced environmental impact.
Without PRRS, producers could have improved herd health, enhanced farm sustainability, reduced strain on farm staff and increased resiliency.

How does PIC breed PRRS-resistant pigs?
To breed pigs that are resistant to PRRS, PIC precisely removed a portion of a specific gene that the PRRS virus enters to infect the pig. Nothing foreign was added. No genes were inserted.
Without that entry point, the PRRS virus cannot infect the pig.
Earning Regulatory Approvals and Protecting Major Global Pork Flows
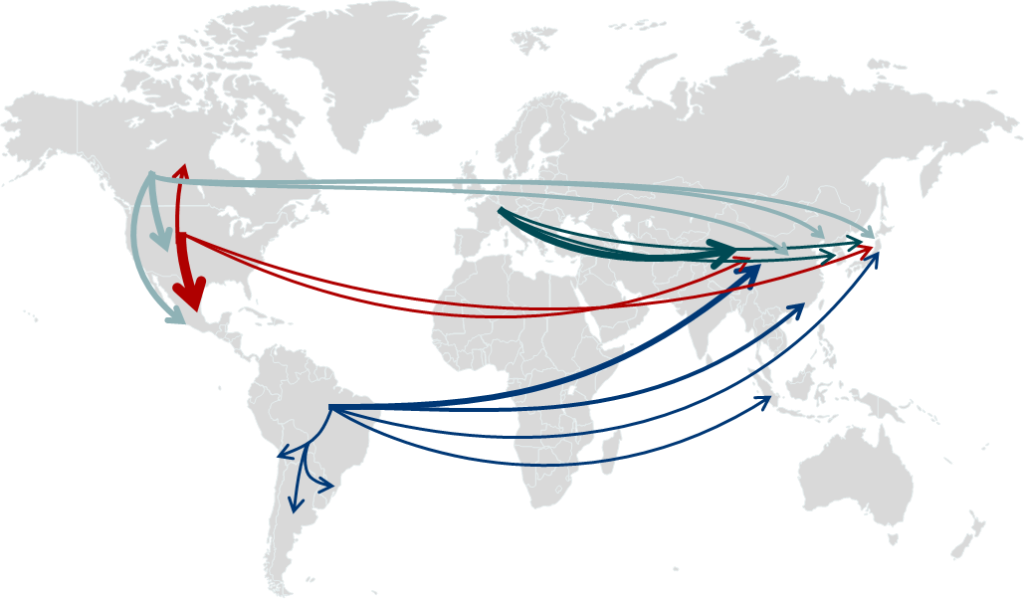
On April 30, 2025, the U.S. Food and Drug Administration (FDA) announced it granted approval to PIC for the gene edit used in its PRRS-resistant pig, determining that the technology is safe and effective.
In 2026, Health Canada and the Canadian Food Inspection Agency (CFIA) approved PIC’s PRRS-resistant pigs for sale as food in Canada after rigorous and thorough review. Environment & Climate Change Canada (ECCC) conducted its own review of the research and data and concluded that PRRS-resistant pigs may now be manufactured or imported in Canada.
Additionally, the governments of Colombia, Brazil, Argentina, Dominican Republic and Uruguay issued favorable regulatory determinations for PRRS-resistant pigs, meaning these countries will treat them the same as any other pigs.
Path to Commercialization in the U.S.
Importantly, FDA approval has not automatically triggered commercialization in the U.S. Though PIC is legally allowed to sell PRRS-resistant pigs in the U.S., we are committed to the responsible and intentional introduction of the PRRS-resistant pig around the globe.
Gaining FDA approval for the technology is an important step in this process, and we are working with additional countries to gain regulatory approval and protect global trade prior to initiating sales and delivery.
Like humans inherit traits from their parents, the pig’s offspring then inherit the PRRS-resistant trait through traditional breeding. That means, even once PIC begins commercializing in the U.S., it will take time to produce enough PRRS-resistant animals to meet industry demand and reach herd immunity. Think of it as a dimmer switch, rather than an on and off switch.
PIC is currently working through commercialization details and will have more information for interested customers soon.

Learn more about PRRS-resistant pig research
Visit PRRSResistantPig.com for more information.



